Keywords
COLLAGEN PLUG
-
Keywords
BiocompatibilityAnalysis of Collagen typeSafety
Technical Data

-
Intended Use
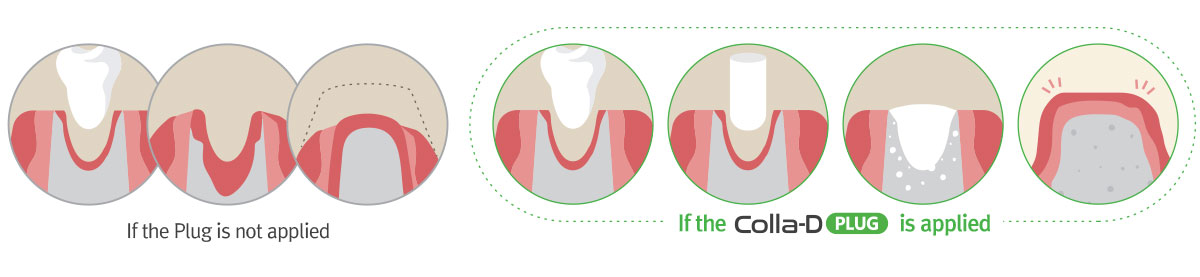
This product is used to induce regeneration of the damaged periodontal
tissue by dental disease or wound, and applied to the local area for dental surgery. -
Preoperative Preparation
1) Check the sterilization status through the sterilization C.I color attached to the product packaging. (Red: good, Yellow: bad)
2) Move the double-packed product to the operating room keeping its sterilization. 3) Check the packaging status of the product and do not use it if the internal packaging is torn or opened.
4) Do not use the product that is expired.
5) Check the label and the content of the product to see if it is suitable for use.
6) Before surgery, the medical team must read the instructions for use of this product and understand the characteristics of the product and how to use it correctly.
7) Use this product as it is or hydrate it using purified water or saline -
Directions for Use
1) Make an incision into the gingiva of the area you want to perform the procedure and expose the periosteum.
2) Completely remove granulation tissue, inflammatory tissue, and other soft tissue from the surgical site.
3) Check the type and size of the product specified on the package and label and remove the product from the package.
4) If necessary, cut the product according to the implantation site using a sterilized apparatus.
5) Insert the product into the application area.
6) Secure the product in the tissue to prevent it from moving and suture the tissue to finish. -
Storage and Expiration date
1) This product shall not be reused as a sterilized disposable medical device. 2) Dispose of products that have been released once, even if they are not used.
-
Precautions
It shall be stored at room temperature (1 to 30°C) and protected from light until use.
Precautions for Use
1. Warning
1) This product shall not be reused as a disposable sterilized medical device. 2) Products that are expired should not be used.
3) This product shall not be used other than a trained dentist or specialist in the relevant business.
2. Cautions for allergies that may occur as a result of the use of medical devices, fatal side effects or accidents caused by careless use of medical devices
1) Patients with collagen allergies
2) Patients with a lesion that has progressed to a point where the residual amount
of alveolar bone has been significantly reduced
3) Patients with a lesion that is difficult to secure space such as horizontal bone defect 4) Patients with multiple dental lesions that require continuous use of multiple
products
5) Uncontrolled diabetics, heavy smokers or alcoholics
6) Patients who lack immune function due to chemotherapy and radiation treatment 7) Patients with intraoral infection or inflammation
(inappropriate oral hygiene or bruxism)
8) Patients with untreatable malocclusion, temporomandibular joint dysfunction and insufficient space in maxillary/mandibular arch
9) Patients Not Suitable for Other Surgery
3. General precautions
1) This product shall not be used for any other purpose than for inducing dental tissue regeneration.
2) Before use, be familiar with all the instructions for use and precautions for use. Infection and side effects may occur if the instruction of use and the precautions for use are not complied with.
3) If side effects occur in patients who have received the transplant, contact the manufacturer immediately and the hospital and doctor who performed the surgery shall take the necessary medical countermeasure for the patient. 4) After opening the package, it is recommended to use this product immediately and take care not to infect the product with external bacteria or microorganisms. 5) Do not use the package if it is damaged or expired.
6) This product shall not be used other than a trained dentist or specialist in the industry.
4. Use for pregnant women, lactating women, fertile women, newborn babies, infants, children, and aged people
Since there is no clinical data for pregnant and lactating women, do not use this product for safety reasons. Do not use the product for immature children.
Manufacturer
MedPark |24, Nakdong-daero 1570 beon-gil, Buk-gu, Busan, Republic of Korea | E-mail : biz@medpark.net
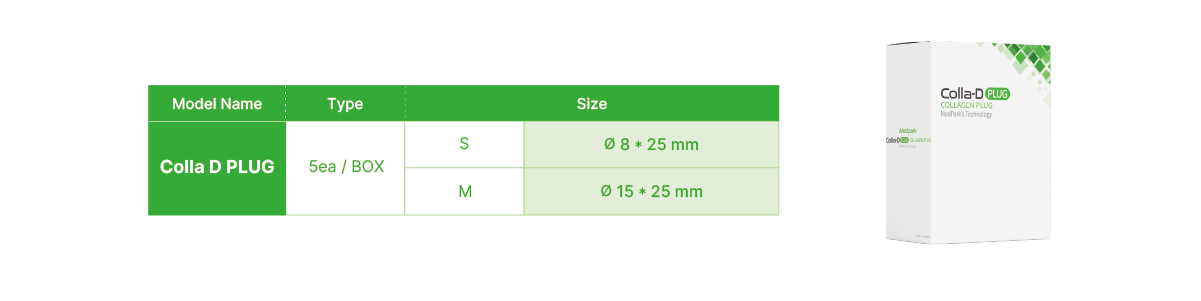
Sepcifications